
The first reason is that the knowledge of p K a of a drug allows predicting the absorption, bio-reactivity and tissular accumulation as a function of the pH of the medium. Also knowing the acidity constant value ( K a) of an acid and its associated p K a value is important for medical students for different reasons. Hence the knowledge of p K a values is useful to the majority of the pharmaceutical companies worldwide. 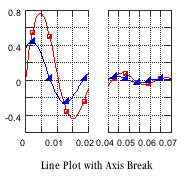
p K a is also closely related to the concept of pH (the acidity of the solution). 
Separation and analytical scientists require an understanding of p K a because it impacts the choice of techniques used to identify and isolate the compound of interest. If a molecule does not readily ionize, it will tend to stay in a non-polar solvent such as cyclohexane (ε 20 = 2) or octanol (ε 20 = 10). Water is a very polar solvent (dielectric constant ε 20 = 80), so facile ionization will increase the likelihood of a species to be taken up into aqueous solution. In biological terms, p K a is important in determining whether a molecule will be taken up by aqueous tissue components or lipid membranes and is related to log P (where P the partition coefficient). p K a is the core property of an electrolyte that defines chemical and biological behavior. Introduction The acid-base ionization/dissociation constant, p K a, is a measure of the tendency of a molecule or ion to keep a proton (H +) at its ionization center(s), and is related to the ionization ability of the acid or base.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
